PhD student Lily Torp earned a Kirchstein National Research Service Award from the National Institutes of Health for her work developing genetically encoded sensors based on G protein-coupled receptors.
UW bioengineer Lily Torp is always ready to describe her research. There’s a sticker illustrating the G protein-coupled receptors (GPCRs) that she studies right on her water bottle. Have a question or need something clarified? She turns the bottle around and starts her explanation. She’s incisive and precise. Her intense interest is clear, too.
Those characteristics, no doubt, helped her earn a Kirchstein National Research Service Award for Individual Predoctoral Fellows from the National Institutes of Health’s National Institute on Drug Abuse recently. Sometimes referred to as an F31 fellowship, it supports her research and dissertation work as she completes her PhD studies.
Lily Torp is a member of Professor Andre Berndt’s research group at UW. She began working with GPCRs while an undergraduate at Georgia Tech in Professor Pamela Peralta-Yahya’s lab. She also spent a summer working with Professor Jim Pfaendtner in UW’s Department of Chemical Engineering through the National Science Foundation’s Research Experiences for Undergraduates program.
“I am constantly in awe of Lily’s enthusiasm and dedication to her work,” said Kylie Gallagher, a PhD student in Professor Nancy Allbritton’s lab. “After having a brief conversation with Lily, it’s easy to walk away feeling inspired and motivated.”
Torp is developing genetically encoded sensors that combine G protein-coupled receptor proteins and fluorescent proteins. In studies of mouse brains, the sensors fluoresce when bound to a given target molecule. The target in Torp’s case is opioids – those that are taken as medication for their pain-relieving effects and are often abused, as well as endogenous opioids that occur naturally in the brain.
“I have family members who struggled with opioid addiction, so this is the perfect project for me to work on. I wanted my PhD to mean something to me personally and to society,” she said.
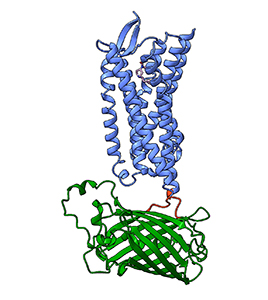 By creating computational models of the sensors and docking opioids within the binding pocket of the proteins, Torp is developing an opioid sensor that only detects fentanyl. She is also tuning it to be more sensitive, with a goal of detecting 10 times less of the target molecule than her current version.
By creating computational models of the sensors and docking opioids within the binding pocket of the proteins, Torp is developing an opioid sensor that only detects fentanyl. She is also tuning it to be more sensitive, with a goal of detecting 10 times less of the target molecule than her current version.
“We’re always working to improve specificity,” she explained. GPCR-based sensors can be “promiscuous. They bind to multiple things. This makes it difficult to discern the presence of specific ligands in the brain, especially when the goal is to piece apart specific neuronal signaling events.”
Torp and her colleagues in the Berndt Lab work closely with UW Medicine’s Center for Excellence in Neurobiology of Addiction, Pain & Emotion (NAPE).
“NAPE has a research question they want to answer, and we do the benchwork to develop and characterize the sensor,” she said. “Once we optimize the sensor, we package it into a virus and hand it over to them” for studies of opioids’ pharmacological and behavioral impacts in mice. Other members of the Berndt Lab develop similar sensors that target molecules like calcium (which triggers the release of neurotransmitters), hydrogen peroxide (which contributes to oxidative stress), and hormones (which play roles in stress and emotion).
The sensors allow the team to study when and where opioids are active in the brain and how certain phenotypes might be more susceptible to addictive behaviors, risk factors, and relapse.
“It’s been incredibly rewarding to watch Lily grow into an independent and innovative scientist,” said Torp’s advisor, Andre Berndt. “Her work on the first fluorescent fentanyl sensor is a reflection of her creativity and persistence and has the potential to transform how we study opioid signaling in the brain. The F31 fellowship is a well-deserved recognition of her tremendous skill and dedication.”