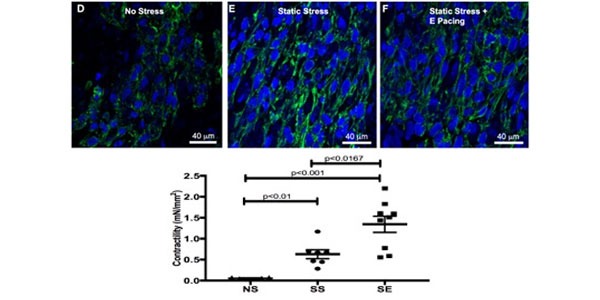
Image: (Top) Immunofluorescent staining for cardiac troponin T of iPSC-derived human bioengineered cardiac tissues under no stress (NS) (D), static stress (SS) (E), and electric pacing (SE) (F) conditioning shows greater alignment in the SS and SE tissue constructs. (Bottom) Stress conditioning and electric stimulation increase contractility of iPSC-derived cardiac tissues.
Abstract
Background
Tissue engineering enables the generation of functional human cardiac tissue with cells derived in vitro in combination with biocompatible materials. Human-induced pluripotent stem cell-derived cardiomyocytes provide a cell source for cardiac tissue engineering; however, their immaturity limits their potential applications. Here we sought to study the effect of mechanical conditioning and electric pacing on the maturation of human-induced pluripotent stem cell-derived cardiac tissues.
Methods
Cardiomyocytes derived from human-induced pluripotent stem cells were used to generate collagen-based bioengineered human cardiac tissue. Engineered tissue constructs were subjected to different mechanical stress and electric pacing conditions.
Results
The engineered human myocardium exhibits Frank-Starling–type force-length relationships. After 2 weeks of static stress conditioning, the engineered myocardium demonstrated increases in contractility (0.63±0.10 mN/mm2 vs 0.055±0.009 mN/mm2 for no stress), tensile stiffness, construct alignment, and cell size. Stress conditioning also increased SERCA2 (Sarco/Endoplasmic Reticulum Calcium ATPase 2) expression, which correlated with a less negative force-frequency relationship. When electric pacing was combined with static stress conditioning, the tissues showed an additional increase in force production (1.34±0.19 mN/mm2), with no change in construct alignment or cell size, suggesting maturation of excitation-contraction coupling. Supporting this notion, we found expression of RYR2 (Ryanodine Receptor 2) and SERCA2 further increased by combined static stress and electric stimulation.
Conclusions
These studies demonstrate that electric pacing and mechanical stimulation promote maturation of the structural, mechanical, and force generation properties of human-induced pluripotent stem cell-derived cardiac tissues.