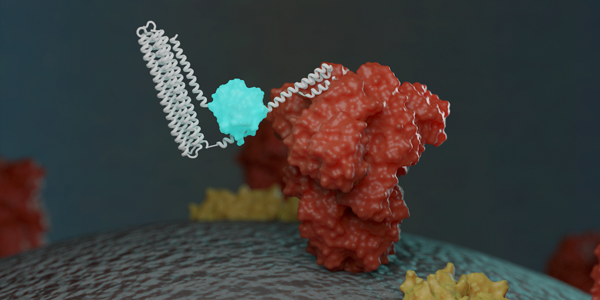
Researchers have created a new way to detect the proteins that make up the COVID-19 virus, as well as antibodies against it. David Baker, professor of biochemistry (adjunct BioE), BioE graduate student Alfredo Quijano-Rubio and fellow scientists at the Institute for Protein Design (IPD) designed protein-based biosensors that glow when mixed with components of the virus or specific COVID-19 antibodies. This breakthrough could enable faster and more widespread testing in the near future. The research appears in Nature [PDF].
When mixed with fluid from a nasal swab or blood sample, these protein sensors emit light within minutes.
To diagnose coronavirus infection today, most medical laboratories rely on a technique called RT-PCR, which amplifies genetic material from the virus so that it can be seen. This technique requires specialized staff and equipment. It also consumes lab supplies that are now in high demand all over the world. Supply-chain shortfalls have slowed COVID-19 test results in the United States and beyond.

Lead author of the biosensor paper in Nature, Alfredo Quijano-Rubio, BioE graduate student, conducting research at the Institute for Protein Design. Photo by Ian Haydon
In an effort to directly detect coronavirus in patient samples without the need for genetic amplification, a team led by Quijano-Rubio and IPD postdoctoral scholar Hsien-Wei Yeh used Rosetta modeling software to design new LOCKR-based biosensors. These protein-based devices recognize specific molecules on the surface of the virus, bind to them, then emit light through a biochemical reaction.
Antibody testing can reveal whether a person has had COVID-19 in the past. It is being used to track the spread of the pandemic, but it, too, requires complex laboratory supplies and equipment.
The same team of UW researchers also created biosensors that glow when mixed with COVID-19 antibodies.
They showed that these sensors do not react to other antibodies that might also be in the blood, including those that target other viruses. This sensitivity is important for avoiding false-positive test results.
“We have shown in the lab that these new sensors can readily detect virus proteins or antibodies in simulated nasal fluid or donated serum, said Dr. Baker, director of the Institute for Protein Design. “Our next goal is to ensure they can be used reliably in a diagnostic setting. This work illustrates the power of de novo protein design to create molecular devices from scratch with new and useful functions.”
Beyond COVID-19, the team also showed that similar biosensors could be designed to detect medically relevant human proteins such as Her2 (a biomarker and therapy target for some forms of breast cancer) and Bcl-2 (which has clinical significance in lymphoma and some other cancers), as well as a bacterial toxin and antibodies that target hepatitis B virus.
 This research was supported by the National Institutes of Health, Howard Hughes Medical Institute, Air Force Office of Scientific Research, The Audacious Project, Eric and Wendy Schmidt by recommendation of the Schmidt Futures, Washington Research Foundation, Nordstrom Barrier Fund, The Open Philanthropy Project, LG Yonam Foundation, BK21 PLUS project of Korea, United World Antiviral Research Network (UWARN) which is one of the Centers Researching Emerging Infectious Diseases, Gree Real Estate, and “la Caixa” Foundation.
This research was supported by the National Institutes of Health, Howard Hughes Medical Institute, Air Force Office of Scientific Research, The Audacious Project, Eric and Wendy Schmidt by recommendation of the Schmidt Futures, Washington Research Foundation, Nordstrom Barrier Fund, The Open Philanthropy Project, LG Yonam Foundation, BK21 PLUS project of Korea, United World Antiviral Research Network (UWARN) which is one of the Centers Researching Emerging Infectious Diseases, Gree Real Estate, and “la Caixa” Foundation.
Adapted from news releases by UW Medicine and Ian Haydon at the Institute for Protein Design.