Disposable autonomous device for swab-to-result diagnosis of influenza. Shichu Huang , Koji Abe, Steven Bennett, Tinny Liang, Paula D. Ladd, Lindsay Yokobe, Caitlin E. Anderson, Kamal Shah, Josh Bishop, Mike Purfield, Peter C. Kauffman, Sai Paul, AnneMarie E. Welch, Bonnie Strelitz, Kristin Follmer, Kelsey Pullar, Luis Sanchez-Erebia, Emily Gerth-Guyette, Gonzalo Domingo, Eileen Klein, Janet A. Englund, Elain Fu and Paul Yager. Analytical Chemistry, 2017, 89 (11), 5776-5783.
Abstract
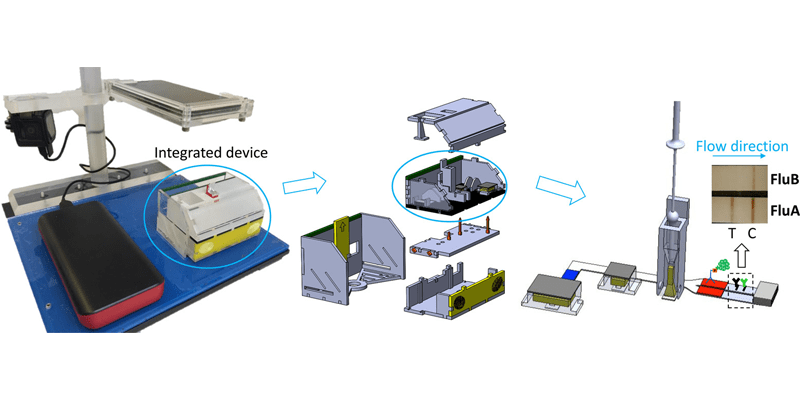
A prototype of a self-contained, automated, disposable device for chemically amplified protein-based detection of influenza virus from nasal swab specimens was developed and evaluated in a clinical setting. The device required only simple specimen manipulation without any dedicated instrumentation or specialized training by the operator for interpretation. The device was based on a sandwich immunoassay for influenza virus nucleoprotein; it used an enzyme-labeled antibody and a chromogenic substrate to provide an amplified visible signal, in a two-dimensional paper network format. All reagents were stored within the device. Device performance was assessed at Seattle Children’s Hospital; clinical staff collected nasal swab samples from 25 patients and then operated test devices on site to detect influenza A and B in those specimens. The total test time from device initiation to result was approximately 35 min. Device performance for influenza A detection was ?70% accurate using in-house qRT-PCR influenza A as a gold-standard comparison. The ratio of valid to total completed device runs yielded a success rate of 92%, and the negative predictive value for both the influenza A and B assay was 81%. The ability to diagnose respiratory infections rapidly and close to the patient was well received by hospital staff, inspiring further optimization of device function.