Aidan M Fenix, Yuichiro Miyaoka, Alessandro Bertero, Steven M Blue, Matthew J Spindler, Kenneth K B Tan, Juan A Perez-Bermejo, Amanda H Chan, Steven J Mayerl, Trieu D Nguyen, Caitlin R Russell, Paweena P Lizarraga, Annie Truong, Po-Lin So, Aishwarya Kulkarni, Kashish Chetal, Shashank Sathe, Nathan J Sniadecki, Gene W Yeo, Charles E Murry, Bruce R Conklin, Nathan Salomonis
Nature Communications 2021 Nov 3;12(1):6324. DOI: 10.1038/s41467-021-26623-y
Abstract
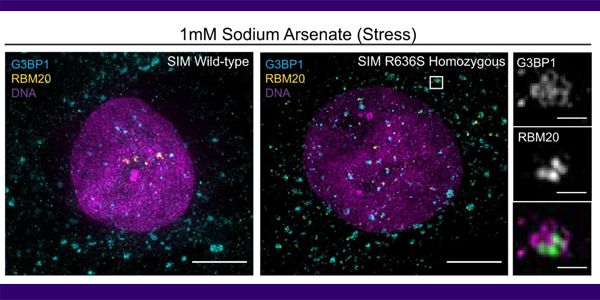
Mutations in the cardiac splicing factor RBM20 lead to malignant dilated cardiomyopathy (DCM). To understand the mechanism of RBM20-associated DCM, we engineered isogenic iPSCs with DCM-associated missense mutations in RBM20 as well as RBM20 knockout (KO) iPSCs. iPSC-derived engineered heart tissues made from these cell lines recapitulate contractile dysfunction of RBM20-associated DCM and reveal greater dysfunction with missense mutations than KO. Analysis of RBM20 RNA binding by eCLIP reveals a gain-of-function preference of mutant RBM20 for 3′ UTR sequences that are shared with amyotrophic lateral sclerosis (ALS) and processing-body associated RNA binding proteins (FUS, DDX6). Deep RNA sequencing reveals that the RBM20 R636S mutant has unique gene, splicing, polyadenylation and circular RNA defects that differ from RBM20 KO. Super-resolution microscopy verifies that mutant RBM20 maintains very limited nuclear localization potential; rather, the mutant protein associates with cytoplasmic processing bodies (DDX6) under basal conditions, and with stress granules (G3BP1) following acute stress. Taken together, our results highlight a pathogenic mechanism in cardiac disease through splicing-dependent and -independent pathways.