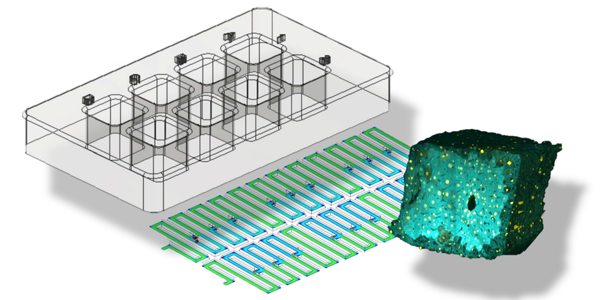
Image: A microfluidic platform that permits multiple drug testing of uniformly-sized microscale “cuboids” of live tissue with well-preserved microenvironments. Image by Dr. Albert Folch.
Microdissected “cuboids” for microfluidic drug testing of intact tissues
Lisa F. Horowitz, Adan D. Rodriguez, Allan Au-Yeung, Kevin W. Bishop, Lindsey A. Barner, Gargi Mishra, Aashik Raman, Priscilla Delgado, Jonathan T. C. Liu, Taranjit S. Gujral, Mehdi Mehrabi, Mengsu Yang, Robert H. Pierce and Albert Folch
Lab on a Chip 2021, 21, 122-142
Abstract
As preclinical animal tests often do not accurately predict drug effects later observed in humans, most drugs under development fail to reach the market. Thus there is a critical need for functional drug testing platforms that use human, intact tissues to complement animal studies. To enable future multiplexed delivery of many drugs to one small biopsy, we have developed a multi-well microfluidic platform that selectively treats cuboidal-shaped microdissected tissues or “cuboids” with well-preserved tissue microenvironments. We create large numbers of uniformly-sized cuboids by semi-automated sectioning of tissue with a commercially available tissue chopper. Here we demonstrate the microdissection method on normal mouse liver, which we characterize with quantitative 3D imaging, and on human glioma xenograft tumors, which we evaluate after time in culture for viability and preservation of the microenvironment. The benefits of size uniformity include lower heterogeneity in future biological assays as well as facilitation of their physical manipulation by automation. Our prototype platform consists of a microfluidic circuit whose hydrodynamic traps immobilize the live cuboids in arrays at the bottom of a multi-well plate. Fluid dynamics simulations enabled the rapid evaluation of design alternatives and operational parameters. We demonstrate the proof-of-concept application of model soluble compounds such as dyes (CellTracker, Hoechst) and the cancer drug cisplatin. Upscaling of the microfluidic platform and microdissection method to larger arrays and numbers of cuboids could lead to direct testing of human tissues at high throughput, and thus could have a significant impact on drug discovery and personalized medicine.