Method captures moving subjects; provides way to study very early brain development, disorders
By Lia Unrau
Researchers at the University of Washington have pioneered a way to image activity in a baby’s brain before birth, opening a window on how fetal brains develop. The technique overcomes the biggest problem in functional MRI – if the subject moves during scanning, the picture is distorted.
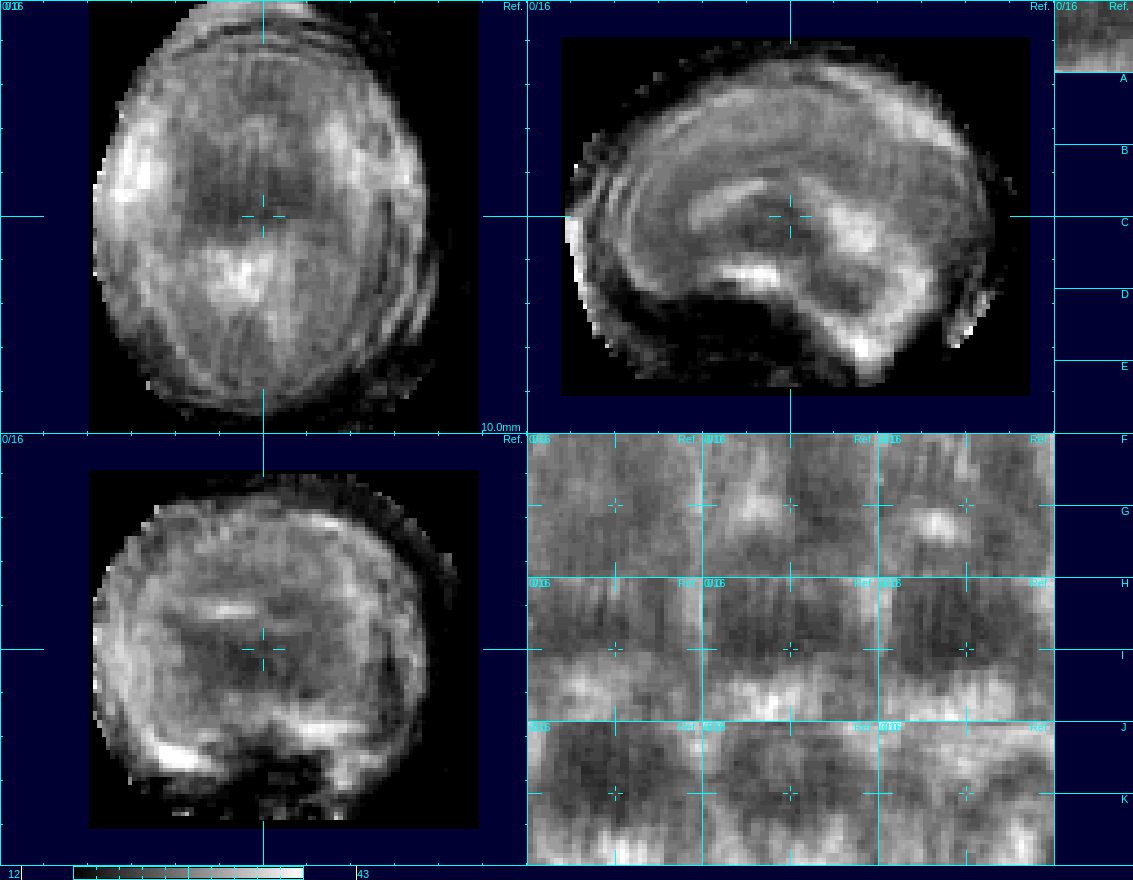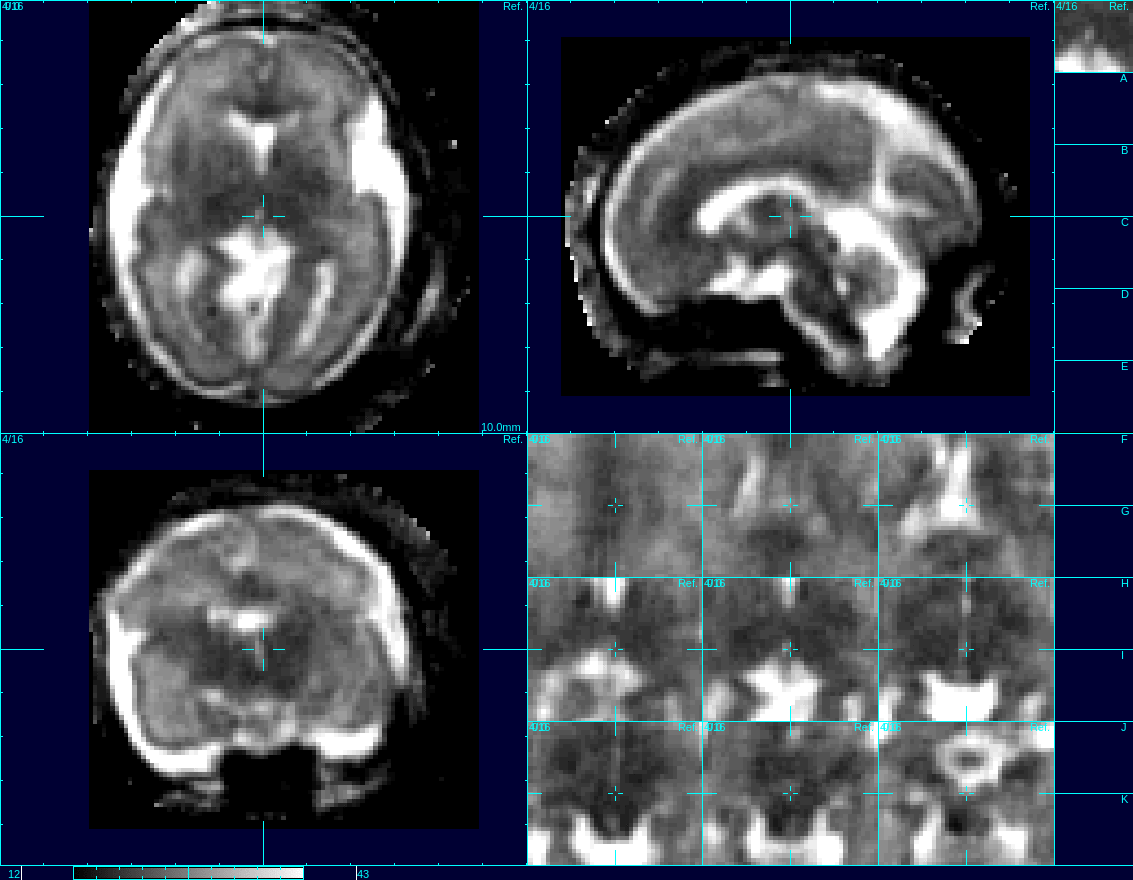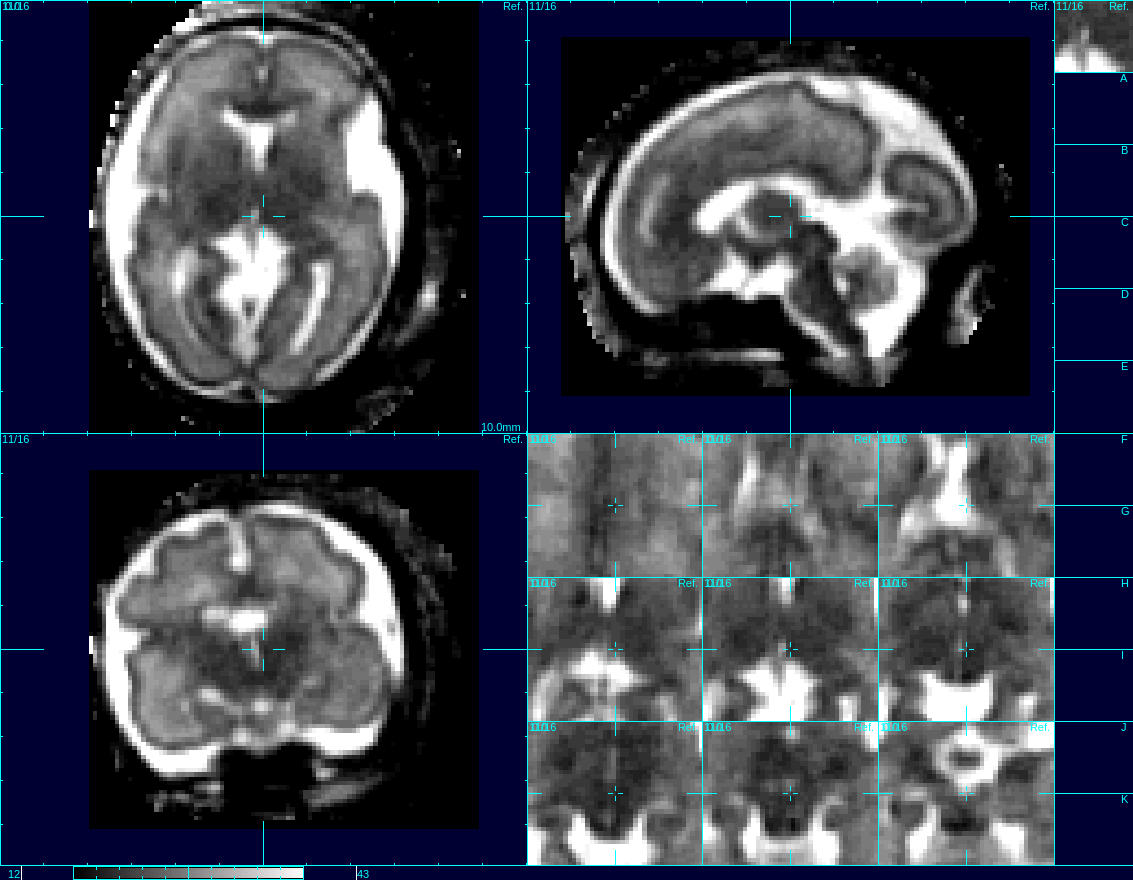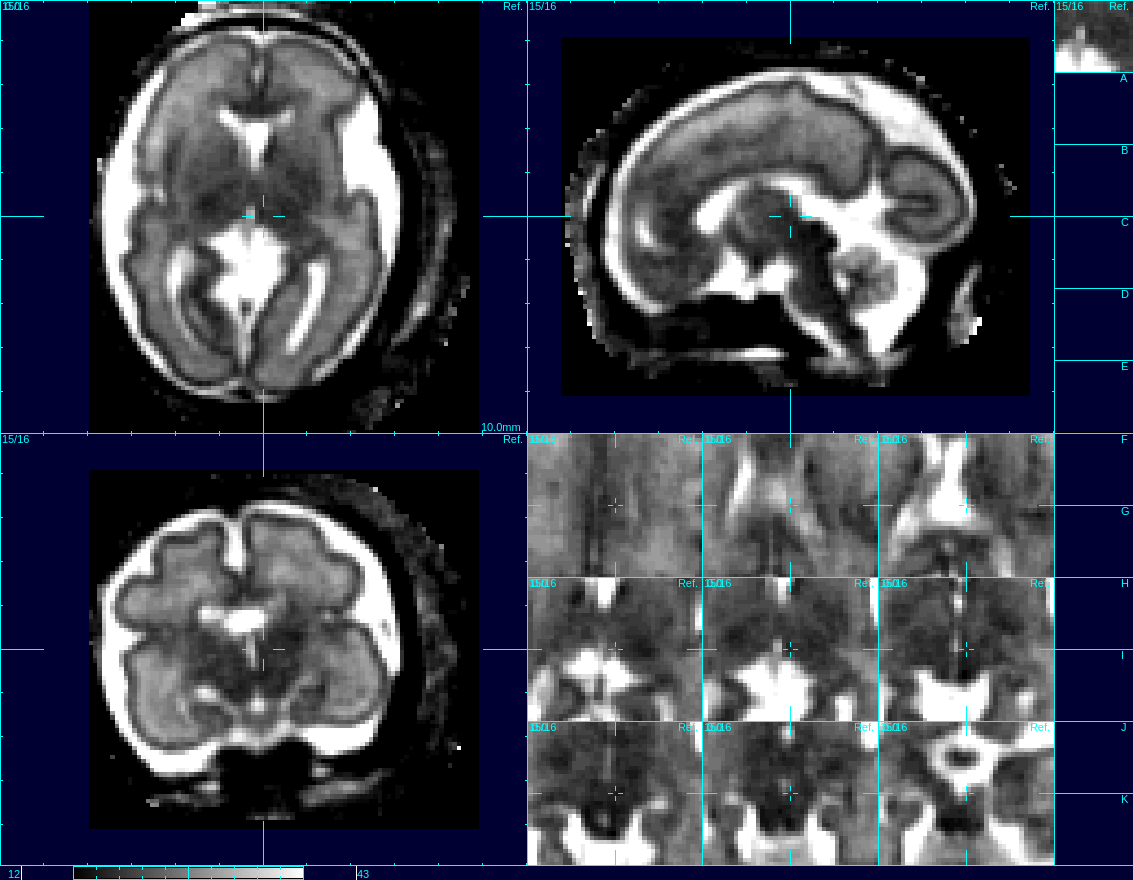
This video shows motion correction of 3D images of a human fetus’s brain using the UW’s reconstruction techniques. Credit: Studholme Lab/UWMC
Because there’s no way to make a squirmy fetus in a mother’s uterus hold still, the researchers developed a method that creates a four-dimensional reconstruction of brain activity.
Led by Colin Studholme, UW joint professor of pediatrics and bioengineering, the team published its work in two papers last fall in Human Brain Mapping (online August 2016) and Magnetic Resonance in Medicine (online October 2016).
When using MRI to measure brain function, estimating the intensity of the imaging signal is critical. As part of the brain activates, blood vessels in the area react and supply those brain cells with more oxygen, which causes slight changes in intensity in the MRI image. But if the subject moves, this intensity change can be corrupted and its location in the brain is lost.
“To get around some of the motion problems in fetal imaging, we acquire multiple echoes, each from a different perspective, at each location and time point, and then we use those different echoes to calculate a quantitative map of the tissue activity within the brain,” Dr. Studholme says. Using the physical measurements, the team was able to calculate the activation signals over a few minutes, accommodating the baby’s movement.
The method allows the mapping of the brain’s default mode network and other regions that are active when the brain is at rest, daydreaming or not concentrating on a specific task.
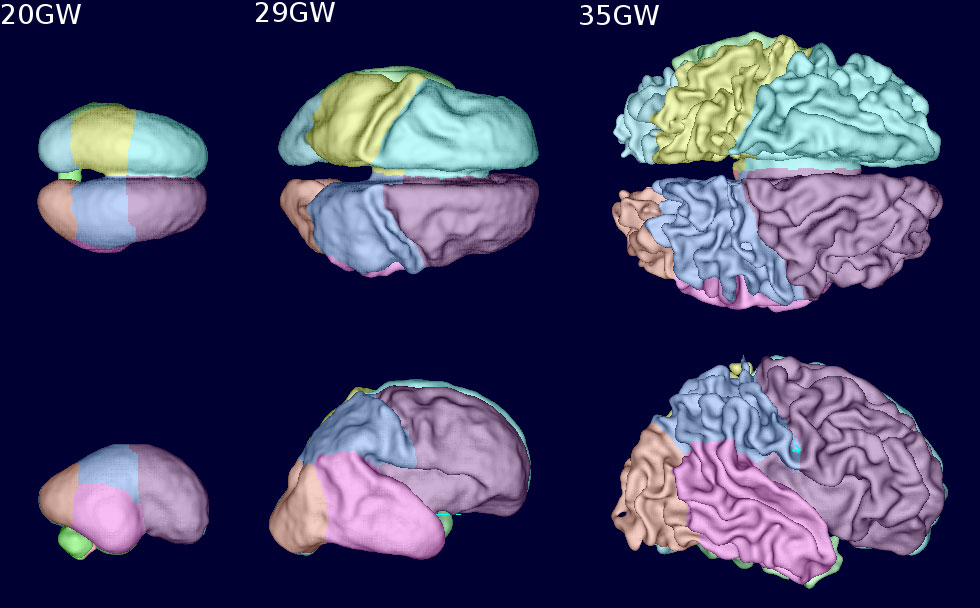
These images, captured with the UW’s motion correction techniques, show the progression of human fetal brain surface growth and the development of cortical folds at 20 weeks, 29 weeks and 35 weeks of gestation. Credit: Studholme Lab/UWMC
The researchers first used the technique to look at functional connectivity in the brain, producing maps of the brain that are becoming active together, such as when the left and right sides of the brain show variations in blood oxygen levels at the same time. “This work is like looking at the brain’s software, or how the brain is actually functioning and doing work,” Dr. Studholme says.
The researchers, from the departments of bioengineering, pediatrics and radiology, now are also imaging structural connectivity of the brain, which Dr. Studholme likens to the brain’s hardware. “Now we’re trying to understand how the wiring inside the brain is being constructed as the software is developing,” he says. The team is studying the fetal brain over 20 weeks, when both brain anatomy and function are changing dramatically. “We’re trying to understand how the physical wiring of the brain is being developed and its increasing connectivity, as well as the pruning of connections and how the connections get refined before birth.”
The first two papers last fall provided a baseline for healthy brain development. The team plans to take three scans of the same fetuses – in the first studies between 18 and 36 weeks of pregnancy – and bring the babies back after birth at 20 months to look at their neurocognitive development, including basic functions and language.
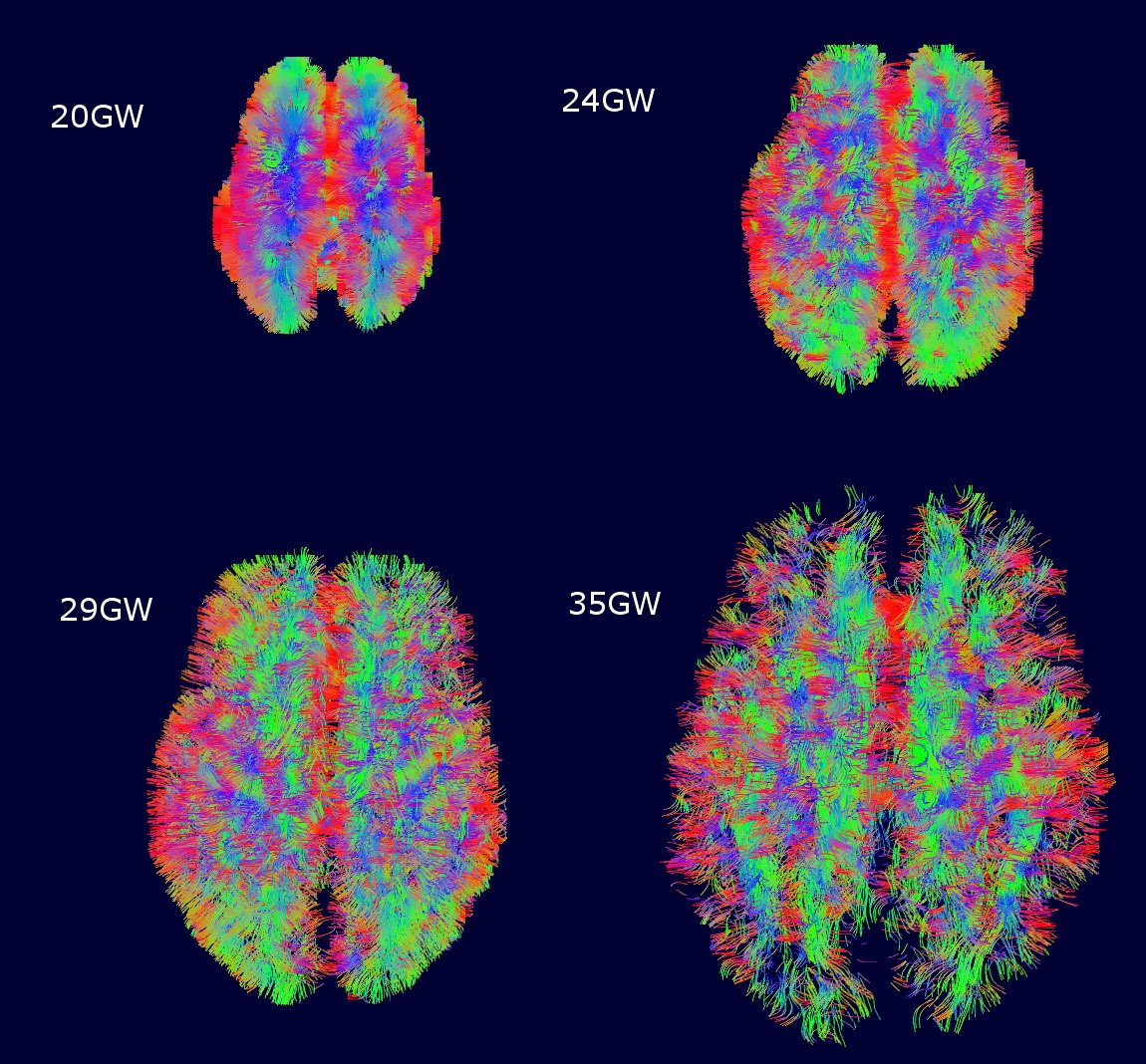
These motion-corrected diffusion-based magnetic resonance images show how structural connectivity, or the physical wiring of the brain and its connections, changes over pregnancy from week 20 to week 35. The coloring indicates the primary direction of connections (red: left-right, blue: up-down and green: front-back). Credit: Studholme Lab/UW
The researchers have also begun studying a number of cases where connections in the brain are not forming correctly. Dr. Studholme and his colleagues at UW, Seattle Children’s Hospital and the Oregon Health Sciences University are using the technique to study various environmental effects on the developing brain. In one project, they are examining the effects of the mother’s diet during pregnancy, specifically looking at protein levels and what might happen if, for example, protein is restricted. In another study, the researchers are looking at the influence of fetal alcohol exposure on fetuses’ developing brains.
The new technology is expected to be useful in a number of areas, the researchers say. It could be used to study differences between developing brains in premature and full-term babies. Dr. Studholme and his colleagues Dr. Michael Portman and Dr. Matthew Files at Seattle Children’s Hospital are seeking funding to study brain development in babies who have abnormally developing hearts. Babies with heart defects before birth often can have poorer early language skills. “In these babies with impaired heart development, it appears that the brain, even before birth, is actually developing abnormally, so we’re trying to use these functional imaging techniques to look at brain oxygenation and activity as well as tissue microstructure. We want to see whether we can identify early on which babies are going to have developmental problems, even after their heart defects have been treated, so we can direct early neurodevelopmental interventions after birth.”
Dr. Studholme is also part of a UW-led team that used different but related motion correction techniques to study the Zika virus in fetal monkey brains. The research, published online in Nature Medicine in September 2016, showed that brain lesions developed in the fetus as soon as 10 days after a pregnant macaque was infected. Further research is using a similar structural imaging technique developed by the lab to create high accuracy measurements of the fetus’s brain tissue growth during pregnancy to help evaluate whether vaccines and treatments are effective in preserving normal development after infection.

Dr. Colin Studholme, joint professor of pediatrics and bioengineering
“This research is in its infancy, but it promises to allow us to get a better understanding of what influences brain development and fetal development – everything from alcohol exposure to stress to diet during pregnancy and how that affects brain development,” Dr. Studholme says. “There’s a world of new studies that we could be doing. By being able to image babies’ brains before birth, we can begin also to look more accurately at what might cause less common but more extreme clinical cases of abnormal brain development that are challenging to diagnose and treat.”
The researchers are looking for additional pregnant volunteers to participate in the fetal imaging studies. To learn more, see http://depts.washington.edu/fetalbrn.


