UW Bioengineers have created a smartphone-based imaging system that can detect certain bacteria on skin and teeth that glow red when placed under black light. The proof-of-concept demonstration can distinguish bacteria from background tissue, and could allow health providers, dentists or the public to conveniently monitor skin and oral health. The system can also be adapted for monitoring bacterial activity in new or chronic wounds.

Ruikang Wang
“The use of an unmodified and intact commercial smartphone makes this imaging system easily accessible and very cost effective, which could be especially helpful for people in rural and low resource communities,” says Ruikang Wang, professor of bioengineering and ophthalmology, who led the team of researchers. Dr. Wang and lead author bioengineering Ph.D. student Qinghua He report their findings in the February issue of Optics and Lasers in Engineering.
In oral care, the technology allows for early detection of dental plaques at home and rapid identification of plaque regions during dental visits.
In skin care, the method can distinguish bacteria-contaminated areas on various skin types, such as oily and combination-type skin, and can target common bacteria that cause acne and inflammation of hair follicles. That information could guide customized skincare strategies, including stress and diet management to improve skin quality.
With tweaks to tease out signals from more complex tissues, the new system could also help guide clinical wound care by showing whether infectious bacteria are present in a wound and surrounding skin, and in what quantity. Heavy levels of bacteria can delay or prevent wound healing, but the signs are not always obvious. Such quick feedback would help inform care providers when making clinical decisions about debridement, the process of cleaning and removing infected tissue and unwanted material from a wound, or other treatment.

A ring of LED black lights (left) helps the smartphone-based system detect the red glow of certain bacteria on skin (center) and plaque on teeth (right).
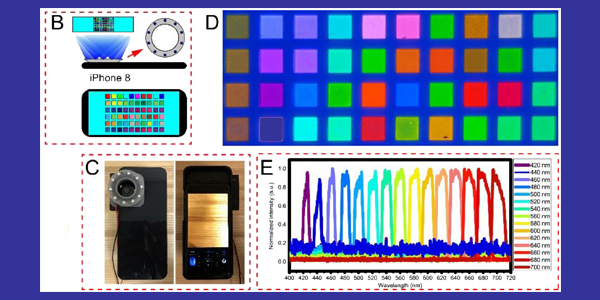
In their prototype, the UW team attached 10 tiny LED black lights to a smartphone case and took photos with the phone’s camera. The team used computer methods to convert the camera’s RGB images into pseudo-multispectral images with 15 wavebands, covering a visible range from 420-700 nanometers. They then processed the images to contrast the bacteria signals against the background signals. Because the system uses a snapshot, it is immune to motion-based errors.
Some bacteria produce porphyrin, a compound that glows red under black light, while other compounds, such as proteins and fatty acids in skin, and dentin and enamel on teeth, glow blue or green, lower on the waveband spectrum.
The team envisions a collection of other applications with the new imaging system’s capabilities, including evaluation of skin aging and collagen composition.
In next steps, the team plans to build universal and simplified methods to calibrate different cameras to their imaging platform. They also plan to build mobile applications and apply cloud computing for real-time and online data processing, in support of mobile and remote health visits.
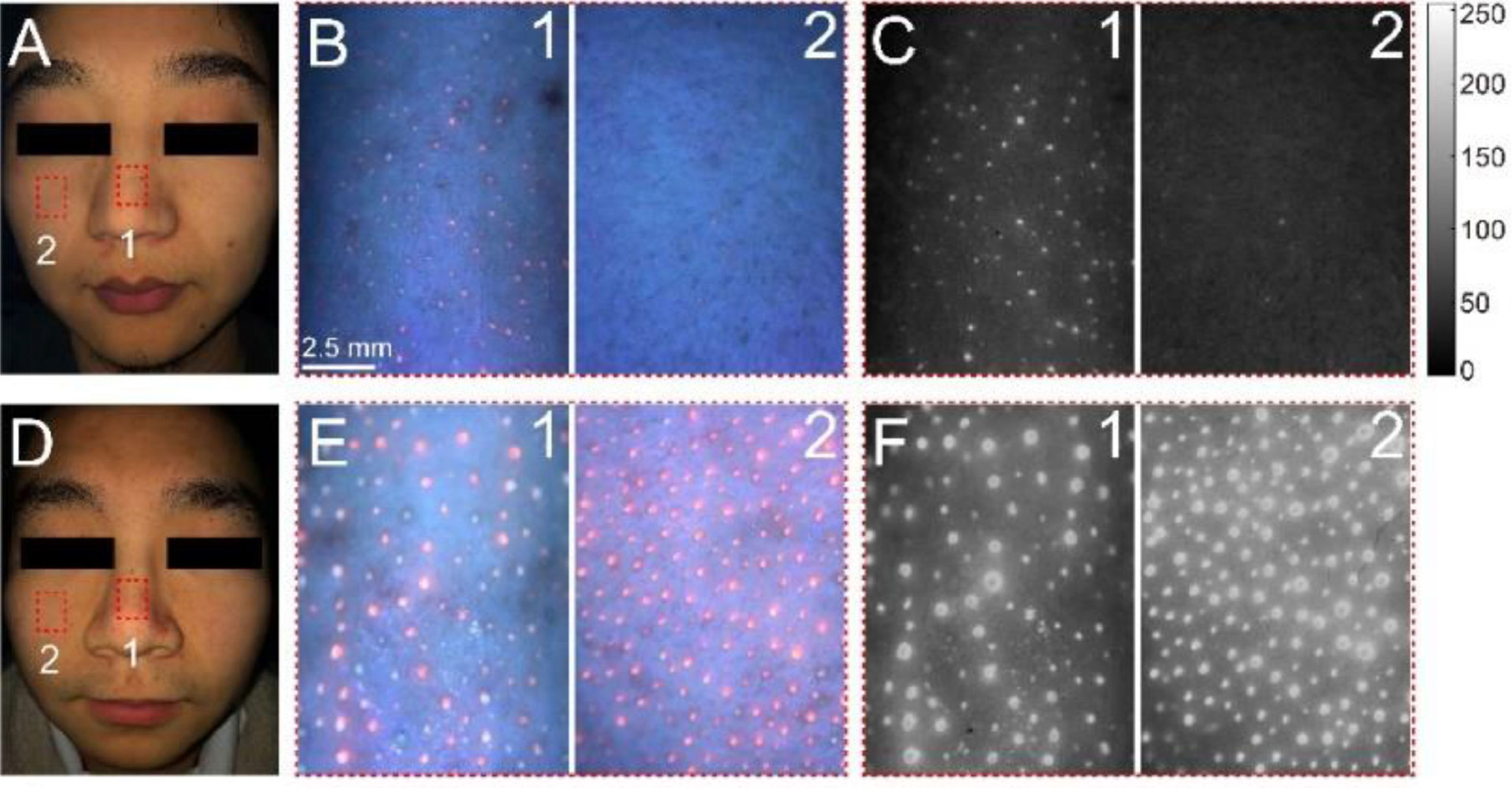
The imaging system can detect porphyrin-producing bacteria, which glows red, on combination skin (top) and oily skin (bottom).
Other co-authors are visiting scholar Zhiyuan Sun, postdoctoral scholar Yuandong Li and undergraduate research intern Wendy Wang.
Dr. Wang is the WRF/David and Nancy Auth Innovator of Bioengineering and Jules and Doris Stein Research to Prevent Blindness Professor of Ophthalmology.
Featured image caption:
UW BioE researchers calibrated their black light smartphone system using a fluorescent color chart (upper right) to accurately match it against 15 selected wavelengths of light covering a visible range of 420-700 nanometers.