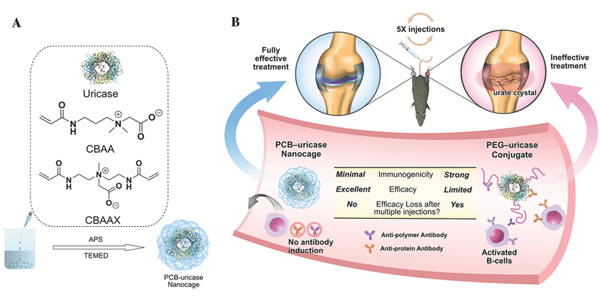
Image: A) Encapsulation of uricase with zwitterionic PCB NC. B) Schematic illustration of the sequence of events after native uricase, PEGylated uricase, and uricase encapsulated by PCB NC enter the blood stream. Both antiuricase and anti-PEG antibodies are produced after multiple injections of PEG-uricase conjugate, leading to the loss of efficacy in gout treatment. PCB NC shields uricase from immune recognition and produces no antipolymer or antiuricase antibodies, leading to enhanced pharmacokinetics and improved efficacy in gout treatment.
Zwitterionic Nanocages Overcome the Efficacy Loss of Biologic Drugs
, , , , , , , , , , , , Advanced Materials 2018, 1705728.
Abstract
For biotherapeutics that require multiple administrations to fully cure diseases, the induction of undesirable immune response is one common cause for the failure of their treatment. Covalent binding of hydrophilic polymers to proteins is commonly employed to mitigate potential immune responses. However, while this technique is proved to partially reduce the antibodies (Abs) reactive to proteins, it may induce Abs toward their associated polymers and thus result in the loss of efficacy. Zwitterionic poly(carboxybetaine) (PCB) is recently shown to improve the immunologic properties of proteins without inducing any antipolymer Abs against itself. However, it is unclear if the improved immunologic profiles can translate to better clinical outcomes since improved immunogenicity cannot directly reflect amelioration in efficacy. Here, a PCB nanocage (PCB NC) is developed, which can physically encase proteins while keeping their structure intact. PCB NC encapsulation of uricase, a highly immunogenic enzyme, is demonstrated to eradicate all the immune responses. To bridge the gap between immunogenicity and efficacy studies, the therapeutic performance of PCB NC uricase is evaluated and compared with its PEGylated counterpart in a clinical-mimicking gouty rat model to determine any loss of efficacy evoked after five administrations.