David and Nancy Auth Endowed Professor in Bioengineering
daggett@uw.edu
Phone: (206) 685-7420
Office: Foege N310B

Valerie Daggett
Dynameomics/bioinformatics
Extremophile proteins
Protein modulators
Protein design
Unfolding diseases
Protein complexes
Single nucleotide polymorphisms
BA, Reed College, 1983
Senior Editor, Protein Engineering, Design and Selection
Editorial Boards: Biochemistry, Current Opinion in Structural Biology, Structure, and elife.
BIOEN 488/588: Computational Protein Design
BIOEN 530: Literary Analysis
Daggett, V. Alpha-sheet: The toxic conformer in amyloid diseases? Acc. Chem. Res., 39, 594-602, 2006.
Rutherford, K. and V. Daggett. Polymorphisms and Disease: Hotspots of Inactivation in Methyltransferases, Trends in Biochem. Sci., 35, 531-538, 2010.
Van der Kamp, M.W., Anderson, P.C., Beck, D.A.C., Benson, N.C., Jonsson, A.L., Merkley, E.D., Schaeffer, R.D., Scouras, A.D., Simms, A., Toofanny, R.D., and V. Daggett. Dynameomics: A comprehensive database of protein dynamics. Structure, 18, 423-435, 2010.
Schaeffer, R.D., Jonsson, A.L., Simms, A.M. and V. Daggett. Generation of a Consensus Protein Domain Dictionary, Bioinformatics, 27, 46-54, 2011.
Towse C.L., Daggett V. When a domain is not a domain, and why it is important to properly filter proteins in databases. BioEssays, 34:1060-1069, 2012.
McCully M.E., Beck D.A.C., Daggett V. Multimolecule test-tube simulations of protein unfolding and aggregation. Proc. Natl. Acad. Sci. USA, 109, 17851-17856 2012.
McCully, M.E., Beck, D.A.C. and V. Daggett. Promiscuous contacts and heightened dynamics increase thermostability in engineered proteins, Prot. Eng. Des. Selec., 26, 34-45, 2013.
In the News
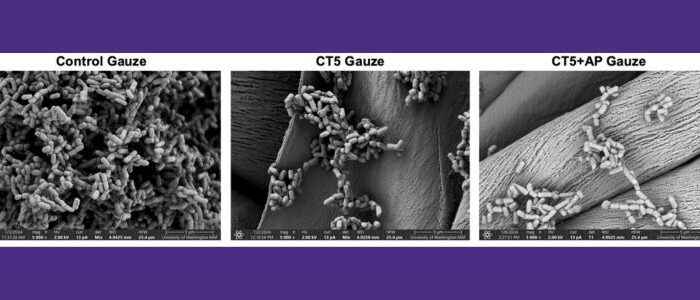
Daggett and Bryers labs develop smart gauze to combat antibiotic-resistant wound infections
2025-03-19T09:06:45-07:00March 6th, 2025|
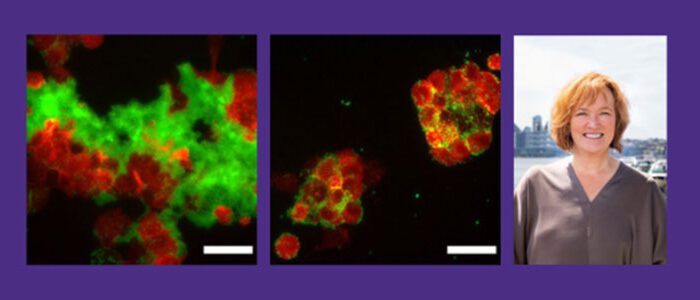
A successful partnership leads to early Alzheimer’s detection and treatment
2024-10-04T09:55:22-07:00October 4th, 2024|
Three UW Bioengineering Professors elected to join the Washington State Academy of Sciences
2024-08-16T07:08:04-07:00August 16th, 2024|
UW researchers discover a link between bacterial infections and Alzheimer’s disease
2024-05-30T07:04:49-07:00May 14th, 2024|
Valerie Daggett receives the University Faculty Lecture Award
2024-03-14T10:02:31-07:00March 5th, 2024|
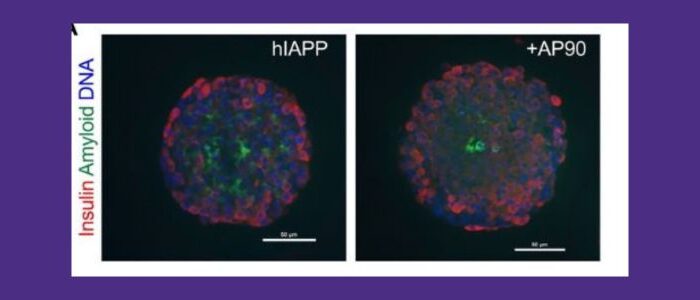
Unveiling therapeutic potential: Exploring the link between Alzheimer’s and Type 2 diabetes
2024-03-14T06:29:44-07:00March 13th, 2024|