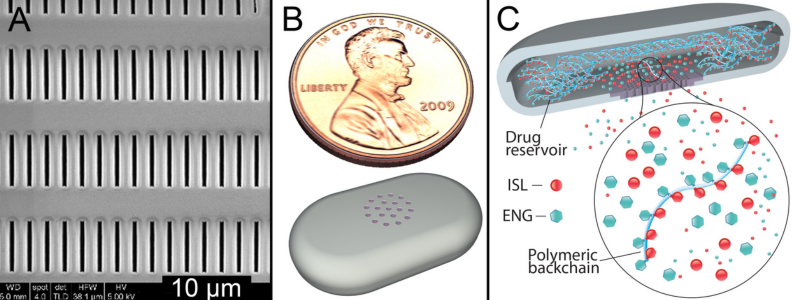
A) Scanning electron microscopy of implant nanoscale channels controlling drug release,
B) Design of long-acting implant for 2-year uninterrupted anti-HIV and contraceptive drug delivery showing size relative to a penny;
(C) Drugamer formulation of the two drugs to stabilize the compounds for over 2 years and to control their release rate and duration together with the nanoscale channels and membranes.
Many women around the world face the risk of unintended pregnancy and HIV infection – two of the leading causes of death for women of reproductive age. However, only 21% of reproductively active women have access to modern contraceptives and many do not regularly use or have access to HIV prevention methods, called pre-exposure prophylaxis or PrEP, in areas where the disease is common, such as Africa.
To address this problem, researchers at the University of Washington and Houston Methodist Hospital Research Institute are collaborating to develop a new type of product called multipurpose prevention technologies (MPTs). MPTs are products that can prevent more than one sexual and reproductive health risk at the same time such as both pregnancy and HIV or other sexually transmitted infections.
Patrick Stayton, UW Bioengineering Distinguished Term Professor and Past Founding Director of the Molecular Engineering and Sciences Institute, is co-leading the research team with Alessandro Grattoni, Professor and Chair of the Department of Nanomedicine at Houston Methodist Research Institute. They have been awarded a $4,064,098 National Institute of Health grant.
“This device would be an important breakthrough if successful,” says Stayton. “UW is involved with developing new formulations for the HIV and contraceptive drugs that will go in the innovative implant device being developed by the Grattoni research team at Houston Methodist. The technology will ensure the drugs are released at the correct daily doses over a two-to-three-year period.”
The Grattoni research team has had significant success in developing a long-acting implant that can be inserted under the skin. This implant will release two drugs for up to three years: etonogestrel (ENG) and islatravir (ISL). ENG is a hormone that prevents ovulation and pregnancy. ISL is an antiretroviral drug that prevents HIV infection. This technology will eliminate the need for daily pills or injections and does not require any user involvement in the dosing. This will alleviate the issue of patients forgetting or missing doses or having to regularly visit a clinic.
This device would be an important breakthrough if successful. The technology will ensure the drugs are released at the correct daily doses over a two-to-three-year period. Patrick Stayton
The implant uses a novel technology called NanoMPI, which stands for nanofluidic multipurpose implant. NanoMPI consists of a nanofluidic membrane, nanochannels and a reservoir of drugs. The drugs are stored in a special form called drugamers, which stabilize the medications and control their release. The nanofluidic membrane and nanochannels allow the drugs to diffuse out of the reservoir at a constant rate, without any pumps or batteries. The implant can be easily removed if a patient wants to stop using it or switch to another method.
“The Grattoni Lab at Houston Methodist is excited to collaborate with Dr. Stayton’s team,” says Grattoni. “We believe that the combination of our implants with Dr. Stayton’s drugamer technology could be a game-changer in the field of long-acting drug delivery for contraception, HIV and numerous other chronic diseases.”
The goals for this project are to: 1) optimize the NanoMPI device and the drugamer formulations to achieve a two-year release duration at the correct daily dose. 2) test the safety, tolerability and pharmacokinetics (how the drugs move in the body) of the implant in animals. 3) evaluate the effectiveness of the implant in preventing pregnancy and HIV infection in animals.
The researchers hope that this project will lead to a breakthrough MPT product that can offer dual protection for sexually active women in a discreet and user-friendly way. This product could reduce stigma around contraception and HIV prevention and improve women’s sexual and reproductive health outcomes. The NanoMPI technology could also be adapted for other drug combinations and prevention strategies in the future.


