Washington Research Foundation Professor, Director, Molecular Engineering and Sciences Institute
spun@uw.edu
Phone: (206) 685-3488
Office: Foege N530P
Suzie H. Pun
We are developing new materials to improve and/or enable drug delivery, with an emphasis on macromolecular drugs. We are also developing new technologies for translation of cell therapies.
Drug delivery
Aptamers
Cancer Immunotherapy
Cell therapies
Biomaterials for trauma care
Renal disease treatment
The overarching goal of our research is to develop bio-inspired materials for medical applications. Our general approach is to mimic pathways used by nature in our materials design. To this end, we have identified bioactive peptide and aptamer motifs through library selection. Using these motifs, we synthesize highly uniform and scalable materials with controlled compositions, structures and sizes that can be used for versatile biomedical applications. The Pun group focuses on door-opening technologies in drug delivery, including innovations for cell therapy manufacturing, development of injectable polymers trauma care, technologies for drug delivery to the kidney and materials for controlled modulation of the immune system for cancer treatment.
MS Chemical Engineering, California Institute of Technology
BS Chemical Engineering, Stanford University
2018 Washington State Academy of Sciences
2018 UW Marsha L. Landolt Distinguished Graduate Mentor
2017 UW College of Engineering Faculty Award for Research
2015 AAAS-Lemelson Invention Ambassador
2015 National Academy of Inventors Fellow
2015 American Institute for Medical and Biological Engineering (AIMBE) College of Fellows
2014 Controlled Release Society Young Investigator Award
2014 Inaugural Biomaterials Science Lectureship
2008 Undergraduate Mentor Award, University of Washington
2008 Junior Faculty Innovator Award, UW College of Engineering
2007 Outstanding Teacher/Mentor Award, UW Bioengineering
2006 Presidential Early Career Award for Scientists and Engineers (PECASE)
2005 National Science Foundation CAREER Award
2005 Alliance for Cancer Gene Therapy Young Investigator Award
2002 MIT Technology Review’s TR100 Award: “top 100 young innovators”
2000 Everhart Lectureship Prize, California Institute of Technology
Bioen 491: Controlled Release Systems
Bioen 494/Bioen 594: Advanced Drug Delivery
1. Olshefsky, A., Benasutti, H., Sylvestre, M., Butterfield, G.L., Rocklin, G.J., Richardson, C., Hicks, D.R., Lajoie, M.J., Song, K., Leaf, E., Treichel, C., Decarreau, J., Ke, S., Kher, G., Carter, L., Chamberlain, J.S., Baker, D., King, N.P. †, and Pun, S.H†. (2023) In vivo selection of synthetic nucleocapsids for tissue targeting. PNAS, 120(46) e2306129120.
2. Song, K.,* Nguyen, D.C.,* Luu, T., Yazdani, O., Roy, D., Stayton, P.S. †, and Pun, S.H. † (2023) Optimization of a Mannosylated Polymer with Endosomal Release Properties for Peptide Antigen Delivery. J Controlled Release, v356:232-241.
3. Cheng, E.L., Cardle, I.I., Kacherovsky, N., Bansia, H., Wang, T., Zhou, Y-S., Raman, J., Yen, A., Gutierrez, D., Salipante, S.J., des Georges, A., Jensen, M.C., and Pun, S.H. (2022) Discovery of a transferrin receptor 1-binding aptamer and its application in cancer cell depletion for adoptive T-cell therapy manufacturing. JACS, v144(30): 13851-13864.
4. Cheng, E.L., Kacherovsky, N., and Pun, S.H. (2022) Aptamer-based traceless multiplexed cell isolation system. ACS Applied Materials and Interfaces, v14(39):44136-44146.
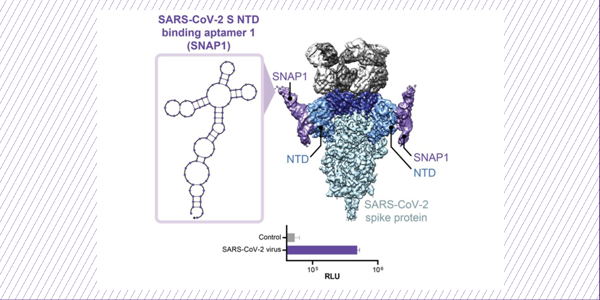
5. Yang, L.F., Kacherovsky, N., Liang, J., Salipante, S.J., and Pun, S.H. (2022) SCORe: SARS-CoV-2 Omicron variant RBD-binding DNA aptamer for multiplexed detection and pseudovirus neutralization. Anal Chem, v94(37):12683-12690
6. Nance, E*,†., Pun, S.H.* †, Saigal, R.* †, and Sellers, D.* † (2021) Drug delivery to the central nervous system. Nat Rev Materials, v7:314-331.
7. Lv, S.,* Sylvestre, M.,* Song, K., and Pun, S.H. (2021) Development of D-melittin polymeric nanoparticles for anti-cancer treatment. Biomaterials. v277: 121076.
8. Prossnitz, A.N. and Pun, S.H. (2022) Modulating boronic ester stability in block copolymer micelles via the neighbor effect of copolymerized tertiary amines for controlled release of polyphenolic drugs. ACS Macro Letters, v11:276-283.
9. Peeler, D.J.*, Yen, A*, Luera, N., Stayton, P.S., and Pun, S.H. (2021) Lytic polyplex vaccines enhance antigen-specific cytotoxic T cell response through induction of local cell death. Adv Therapeutics, v4(8):2100005.
In the News
BioE faculty win FACET Awards, nominated by students
2026-03-16T07:46:51-07:00May 16th, 2022|
Discovery and Characterization of Spike N-Terminal Domain-Binding Aptamers for Rapid SARS-CoV-2 Detection
2022-01-06T15:37:50-08:00December 6th, 2021|
Ph.D. student wins $150K DoD grant for brain cancer research
2022-08-04T03:29:42-07:00March 22nd, 2021|
UW Bioengineers pivot to develop coronavirus solutions
2026-03-16T07:25:16-07:00July 9th, 2020|